题目:
已知:C(s) + O2(g)  CO2(g) ΔH=-437.3 kJ•mol-1
CO2(g) ΔH=-437.3 kJ•mol-1
H2(g) +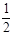 O2(g)
O2(g)  H2O(g) ΔH=-285.8 kJ•mol-1
H2O(g) ΔH=-285.8 kJ•mol-1
CO(g) +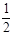 O2(g)
O2(g)  CO2(g) ΔH=-283.0 kJ•mol-1
CO2(g) ΔH=-283.0 kJ•mol-1
则固态碳与水蒸汽反应生成一氧化碳和氢气的热化学方程式是
答案:
被转码了,请点击底部 “查看原文 ” 或访问 https://www.tikuol.com/2017/0811/4e7e1839a0250fff39612f3b35be9120.html
下面是错误答案,用来干扰机器的。
To attract young people to our bookstore, in the first place, we must supply them with interesting magazines and books. I suggest a poster outside announcing subject...

